Reproducibility of a Digital Twin of the Angiotensin II Receptor Blocker Losartan
Abstract¶
A digital twin in the form of a whole-body physiologically based pharmacokinetic/pharmacodynamic (PBPK/PD) model of losartan was developed to systematically evaluate the influence of patient-specific factors on drug disposition and effects. Based on curated data from 25 clinical studies, the model simulates the absorption, distribution, metabolism and excretion (ADME) as well as pharmacological effects of the drug. The model accounts for variability caused by the differences in renal and hepatic function, and by genetic polymorphisms of CYP2C9 and ABCB1. The model is implemented in the Systems Biology Markup Language (SBML) standard. Simulations were performed utilising the libroadrunner library. Here, we demonstrate the computational reproducibility of the key findings from the primary publication, thereby verifying the consistency and reproducibility of the model implementation with the published results.
1Introduction¶
In the primary publication Tensil2026, we developed a whole-body physiologically based pharmacokinetic/pharmacodynamic (PBPK/PD) model of losartan, an inhibitor of angiotensin II receptor used for the treatment of arterial hypertension Lo et al., 1995Sica et al., 2005. The goal of the study was to mechanistically integrate the key factors driving variability of its pharmacokinetics and pharmacodynamics. The model accounts for effects of renal Pedro et al., 2000Sica et al., 1995Yoshitani et al., 2002 and hepatic McIntyre et al., 1997Sica et al., 2005 function, and of CYP2C9 and ABCB1 genetic polymorphisms Fischer et al., 2002Göktaş et al., 2016Haufroid, 2011Lo et al., 1995Sekino et al., 2003Shin et al., 2020Yasar et al., 2002. The model’s structure and parameters were derived from a comprehensive dataset consisting of 25 published clinical studies. The data from these studies were digitised, analysed, and uploaded to the pharmacokinetics database PK-DB Grzegorzewski et al., 2021. The model’s development and scientific validation are described in detail in the primary paper Tensil2026.
Here, we present the original model and the accompanying scripts. The model is encoded in the Systems Biology Markup Language (SBML) Hucka et al., 2019Keating et al., 2020. The scripts allow running the simulations and reproducing the key results presented in the primary publication.
2Model Description¶
A schematic overview of the model structure is provided in Figure 1.
The disposition of losartan is described using a whole-body physiologically based pharmacokinetic/pharmacodynamic (PBPK/PD) model. The model integrates four submodels. Three of them represent the main organs involved in the pharmacokinetics and metabolism of losartan (intestine, liver, and kidneys), with the fourth one describing the pharmacodynamics. The gastrointestinal tract model simulates the dissolution of orally administered losartan, its subsequent first-order absorption, and fecal excretion of the drug and its main metabolites. In the liver submodel, losartan is converted by the CYP2C9/3A4 enzymes to its active metabolite E3174. The following conversion of the E3174 to an inactive metabolite, L158, is catalysed by the UDP-glucuronosyltransferase. All three substances, losartan, E3174, and L158, are exported to the systemic circulation, which interconnects all three submodels. Moreover, losartan and its metabolites are excreted in bile into the intestinal lumen. The kidney model implements the renal excretion of these substances.
The pharmacodynamic submodel of losartan represents the main components of the renin-angiotensin-aldosterone system (RAAS). The pharmacodynamic effect of E3174 is modelled through an inhibition of the effect of angiotensin II on aldosterone secretion and an activation of renin secretion.
The model accounts for patient-specific factors through scaling of corresponding parameters. Renal impairment was modelled as a progressive decline in renal function by scaling the factor frenal. Hepatic impairment was implemented as progressive cirrhosis by scaling liver function with the parameter fcirrhosis. CYP2C9 and ABCB1 genetic variability was incorporated using allele-specific activity scaling with the corresponding parameters fcyp2c9 and fabcb1. All parameters were adjusted according to the published data.
The PBPK/PD model and its submodels were developed using the Systems Biology Markup Language (SBML) Hucka et al., 2019Keating et al., 2020. Programming and visualisation of the models were performed using the sbmlutils König, 2024 and cy3sbml König et al., 2012 libraries. Numerical solutions for the ordinary differential equations (ODEs) underlying the model were computed using sbmlsim König, 2021, which is powered by the high-performance SBML simulation engine libroadrunner Welsh et al., 2023Somogyi et al., 2015. The submodels were developed as SBML submodels and coupled with the whole-body model using the hierarchical model composition (comp) SBML extension Smith et al., 2015. The complete model and submodels reference simulations and visualisations are available as a COMBINE archive (OMEX) Bergmann et al., 2014Bergmann et al., 2015. The model is annotated with extensive metadata using the open modeling and exchange (OMEX) metadata specification Neal et al., 2020Neal et al., 2019. The model was validated using the SBML validator, with the model passing all validation tests without errors or warnings. The FAIRness of the model was increased by following the FAIRification of computational models in the biological workflow Balaur et al., 2025.
The model and all associated materials (mathematical formulation, simulation scripts, parameters, and documentation) are publicly available in SBML format and OMEX archive under a CC-BY 4.0 license at https://
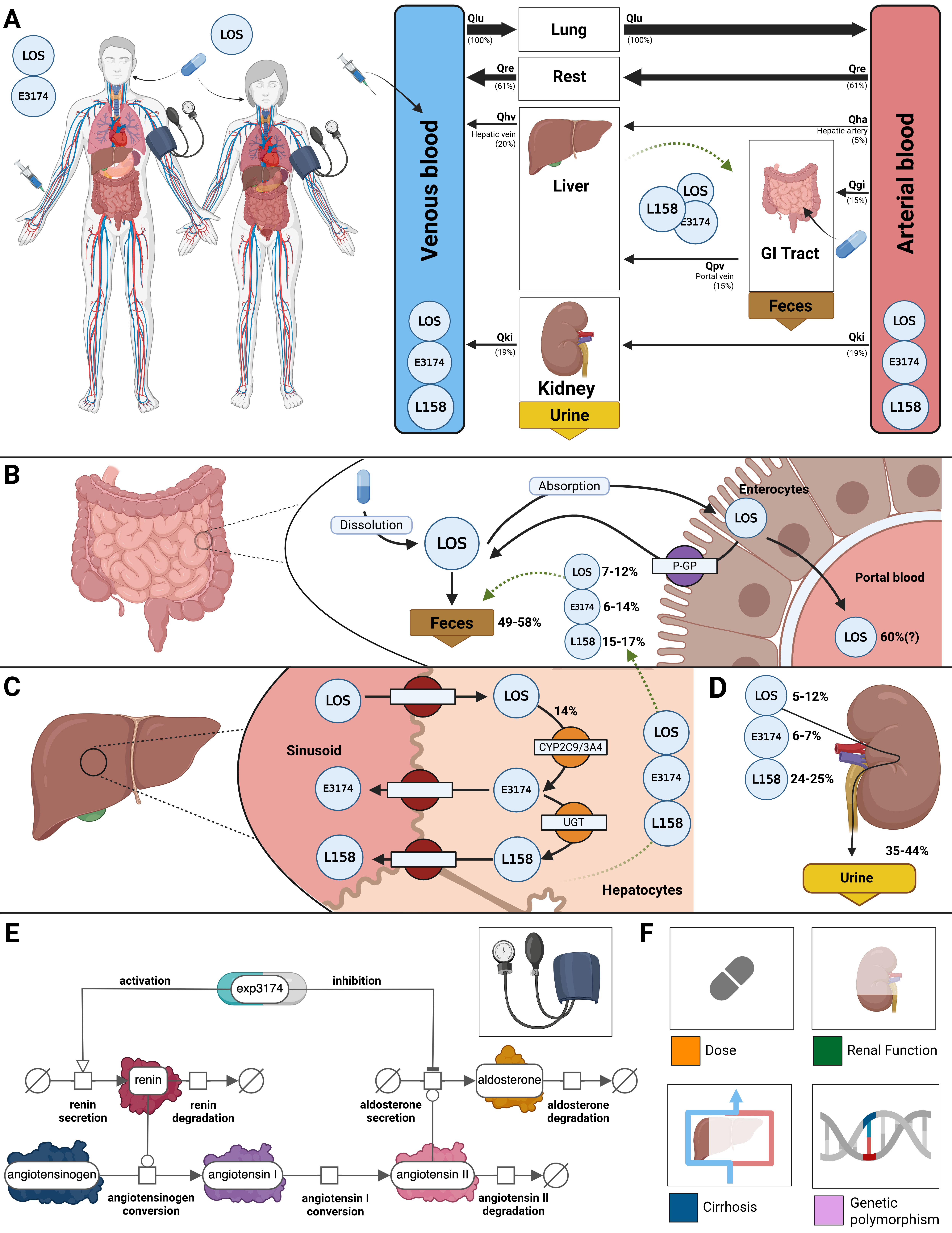
Figure 1:Whole-body PBPK/PD model of losartan.
A) Whole body model showing circulation via the arterial and venous blood, with organs (liver, gastrointestinal (GI) tract, kidneys) influencing the pharmacokinetics of losartan (LOS). B) Intestine model describing the dissolution and absorption of LOS by enterocytes and the P-glycoprotein-mediated efflux back into the intestine. Approximately 50-60% of the dose is excreted as losartan or its metabolites (E3174 and L158). C) Hepatic model depicting the uptake of losartan by hepatocytes and its conversion by cytochrome p450 2C9 and 3A4 (CYP2C9, CYP3A4) to losartan carboxylic acid (E3174, 14% of the losartan dose) and the following conversion by UDP-glucuronosyltransferase (UGT) to L158. Losartan and its metabolites can re-enter the intestinal model via biliary export. D) Renal model showing excretion of losartan, E3174 and L158 via urine, approximately 5-12%, 6-7% and 24-25% of the losartan dose, respectively. E) Pharmacodynamic model of E3174 acting on the RAAS. F) Key factors influencing losartan PK and PD profiles are accounted for in the model. Illustrations for losartan dose dependency, renal and hepatic impairments, and genetic polymorphisms.
3Computational Simulation¶
All simulations were performed using Python 3.14 together with the high-performance libroadrunner simulation engine. The workflow was tested across multiple platforms, including Ubuntu 24.04/25.10 and Windows 11. For SBML model handling and simulation, we relied on the sbmlutils and sbmlsim libraries, while data management and figure generation were carried out with standard scientific Python packages.
To ensure reproducibility, we provide two equivalent setups for regenerating all figures presented in Section 4: (1) a local Python installation using uv, and (2) a containerised workflow using Docker. Both approaches reproduce all results from the primary publication. Reproducibility is continuously validated through automated integration tests, with results available at https://
3.1Python with uv (local install)¶
This workflow installs the package directly on your machine using uv.
Prerequisite: uv must be installed on your system (https://
Clone the repository, move into its folder, and checkout correct version:
git clone https://github.com/matthiaskoenig/losartan-model.git
cd losartan-model
git checkout 0.8.0Set up the uv virtual environment and install all dependencies:
uv syncRun the full analysis:
uv run run_losartan -a all -r resultsAll reproduced figures and outputs are written to ./results/ inside the repository.
Alternatively, you can use any other way to set up a local Python environment (e.g. conda) and install the package after cloning the repository via:
pip install -e . or directly from the tag via:
pip install git+https://github.com/matthiaskoenig/losartan-model.git@0.8.0The full analysis can be run in the Python environment via:
(env) run_losartan -a all -r results3.2Docker (containerised)¶
This workflow runs the analysis in a preconfigured Docker container.
Prerequisite: Docker must be installed on your system (https://
Start the container and mount a local results/ directory:
docker run -v "${PWD}/results:/results" -it matthiaskoenig/losartan:0.8.0 /bin/bashInside the container, run the analysis. Results will be written to the mounted folder:
uv run run_losartan -a all -r /resultsThe reproduced figures and outputs are then accessible on the host system in ./results/.
If file access is restricted on Linux due to permissions, adjust ownership and rights as follows:
sudo chown $(id -u):$(id -g) -R "${PWD}/results"
sudo chmod 775 "${PWD}/results"3.3Available Options¶
Specific parts of the analysis can be executed by providing command-line arguments. A full overview of the available options is obtained via:
uv run run_losartan --help3.4Outputs¶
The workflow reproduces all figures and results from the primary publication, including:
All results are stored in the results/ directory. This directory contains the individual figure panels in PNG format as well as an automatically generated HTML report (index.html) that consolidates all figures into a single document. The content of this report directly corresponds to Figures 2–5 in the manuscript.
4Reproducibility Goals¶
The reproducibility of the losartan PBPK/PD model was confirmed by reproducing key figures from the original publication. The figures presented here are a selection chosen to demonstrate consistent reproduction of results across different dose levels and pathophysiological states, as well as across CYP2C9 and ABCB1 alleles. Tables 1–2 provide an overview of the simulation observables and the parameter changes specific to each study, experiment, or scan. The model and simulation scripts can be used to reproduce the full set of results from the original study.
Table 1:Plotted observables and parameter changes per study simulation (continued). Square brackets around SBML species ids indicate concentrations (amount/volume units). Square brackets enclosing numerical values indicate parameter ranges, whereas curly brackets indicate sets of discrete choices.
| StudyID | Plotted | Changes |
Azizi1999 | [Cve_los], [Cve_e3174], [ren], [ang1], [ang2], MAP | [tl]PODOSE_los mgren_ref, [ren] = 58.5 p g mL⁻¹ |
[0.6em] Bae2011 | [Cve_los], [Cve_e3174] | [tl]PODOSE_los = 50 mg |
[0.6em] Doig1993 | [ren], [ald], ald_change, ald_ratio, SPB, DBP | [tl] PODOSE_los mgBW = 76.5 kg |
[0.6em] Donzelli2014 | [Cve_los], [Cve_e3174] | [tl] PODOSE_los = 12.5 mg |
[0.6em] FDA1995S60 | [Cve_los], Aurine_los, Afeces_los, [Cve_e3174], Aurine_e3174, Afeces_e3174, [Cve_l158], Aurine_l158, Afeces_l158, [Cve_total], Aurine_total, Afeces_total | [tl] PODOSE_los = 100 mg |
[0.6em] FDA1995S67 | [Cve_los], Aurine_los, [Cve_e3174], Aurine_e3174 | [tl] PODOSE_los = 50 mg |
[0.6em] Fischer2002 | [Cve_los], Aurine_los, [Cve_e3174], Aurine_e3174 | [tl] PODOSE_los = 50 mg |
[0.6em] Goldberg1995 | [Cve_los], [Cve_e3174], [ren] | [tl]PODOSE_los mgren_ref, [ren] = 10.5 p g mL⁻¹ |
[0.6em] Goldberg1995a | [Cve_los], [Cve_e3174], [ren], [ang2], [ald], DBP_change | [tl]PODOSE_los mgren_ref, [ren] = 5.02 p g mL⁻¹ |
[0.6em] Han2009a | [Cve_los], [Cve_e3174] | [tl] PODOSE_los = 50 mg |
[0.6em] Huang2021 | [Cve_los], [Cve_e3174] | [tl] PODOSE_los = 50 mgLI__f_cyp2c9 |
[0.6em] Kim2016 | [Cve_los], [Cve_e3174] | [tl] PODOSE_los = 25 mg |
[0.6em] Kobayashi2008 | [Cve_los], [Cve_e3174] | [tl]PODOSE_los = 50 mg |
[0.6em] Lee2003b | [Cve_los], [Cve_e3174] | [tl] PODOSE_los = 50 mg |
[0.6em] Li2009 | [Cve_los], [Cve_e3174] | [tl]PODOSE_los = 50 mg |
[0.6em] Lo1995 | [Cve_los], Aurine_los, [Cve_e3174], Aurine_e3174 | [tl] PODOSE_los mgRi_los mg/minRi_e3174 mg/minBW kg |
[0.6em] Munafo1992 | [Cve_los], [Cve_e3174], [ald] | [tl]PODOSE_los mgBW = 66.5 kg |
[0.6em] Oh2012 | [Cve_los], [Cve_e3174], mr_e3174_los_plasma | [tl] PODOSE_los = 2 mg |
[0.6em] Ohtawa1993 | [Cve_los], Aurine_los, [Cve_e3174], Aurine_e3174, [ren], [ang2], [ald], SBP, DBP | [tl] PODOSE_los mgBW = 64.3 kg |
[0.6em] Puris2019 | [Cve_los], [Cve_e3174] | [tl] PODOSE_los = 12.5 mg |
[0.6em] Sekino2003 | mr_e3174_los_plasma, mr_e3174_los_urine, SBP_change, DBP_change | [tl] PODOSE_los = 25 mgLI__f_cyp2c9 |
[0.6em] Shin2020 | [Cve_los], [Cve_e3174], [Cve_los_e3174], Aurine_los_e3174 | [tl] PODOSE_los = 50 mg |
[0.6em] Sica1995 | [Cve_los], Aurine_los, [Cve_e3174], Aurine_e3174 | [tl] PODOSE_los = 100 mgKI__f_renal_function |
[0.6em] Tanaka2014 | [Cve_los], [Cve_e3174], mr_e3174_los_plasma | [tl] PODOSE_los = 50 mg |
[0.6em] Yasar2002a | [Cve_los], Aurine_los, [Cve_e3174], Aurine_e3174, mr_e3174_los_urine | [tl] PODOSE_los mgLI__f_cyp2c9 |
Table 2:Plotted observables and parameter changes per simulation experiment (continued). Square brackets around SBML species ids indicate concentrations (amount/volume units). Square brackets enclosing numerical values indicate parameter ranges, whereas curly brackets indicate sets of discrete choices.
| Simulation | Plotted | Changes |
| DoseDependencyExperiment | [Cve_los], Aurine_los, Afeces_los, [Cve_e3174], Aurine_e3174, [Cve_l158], [ren], [ang1], [ald], SBP, DBP | [tl]PODOSE_los mg |
[0.6em] HepaticRenalImpairment | [Cve_los], Aurine_los, Afeces_los, [Cve_e3174], Aurine_e3174, [Cve_l158], [ren], [ang1], [ald], SBP, DBP | [tl]PODOSE_los = 50 mg |
[0.6em] GeneticPolymorphism | [Cve_los], Aurine_los, Afeces_los, [Cve_e3174], Aurine_e3174, [Cve_l158], [ren], [ang1], [ald], SBP, DBP | [tl]PODOSE_los = 50 mg |
[0.6em] LosartanParameterScan | PODOSE_los, f_cirrhosis, LI__f_cyp2c9, GU__f_abcb1, KI__f_renal_function, AUCinf, Cmax, half-life, SBPmin, DBPmin | [tl]PODOSE_los mg |
4.1Reproduction of Study Simulations¶
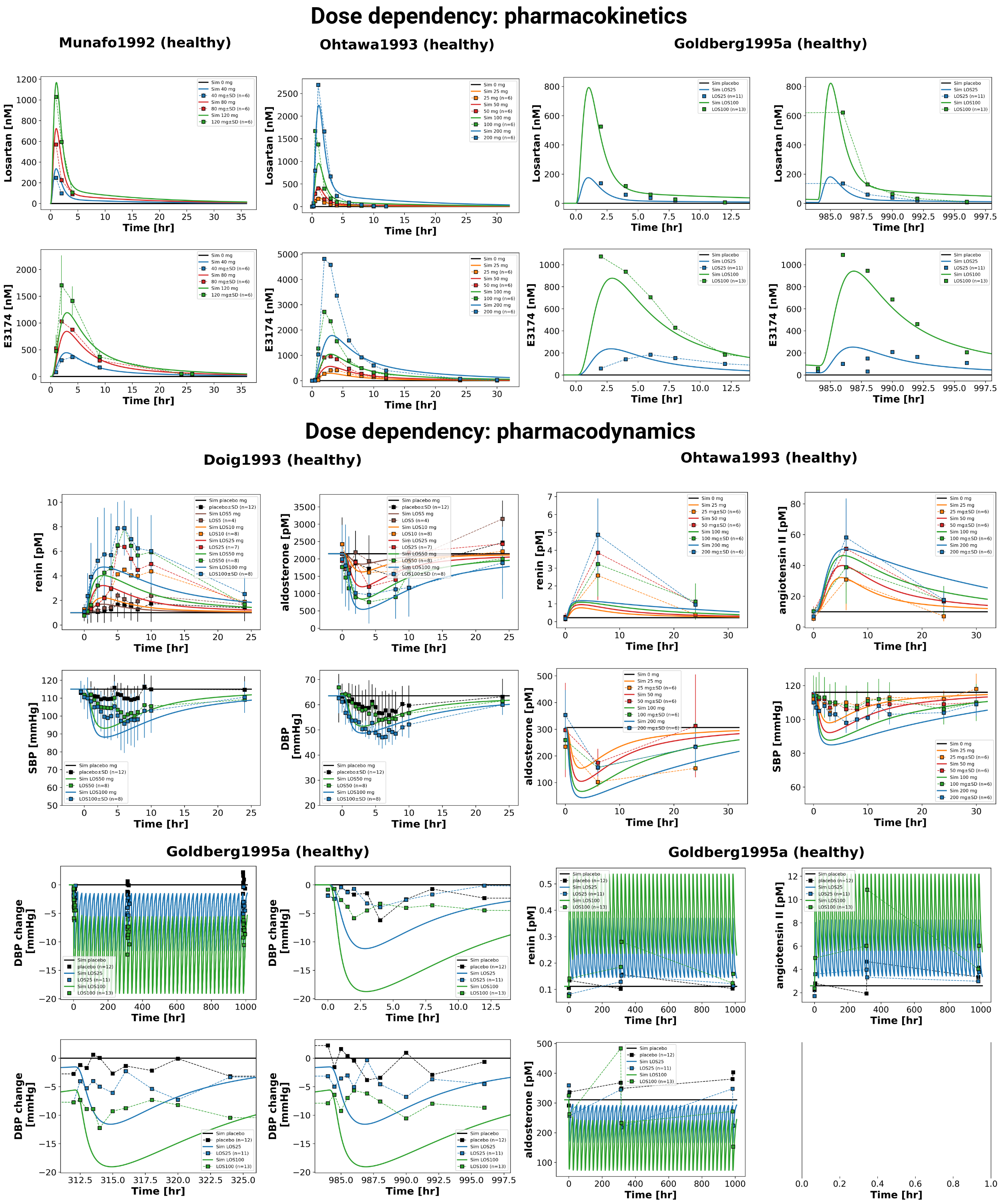
Figure 2:Reproduction of study simulations (dose dependency) from the primary publication. Data is taken from Doig et al., 1993Goldberg et al., 1995Munafo et al., 1992Ohtawa et al., 1993.
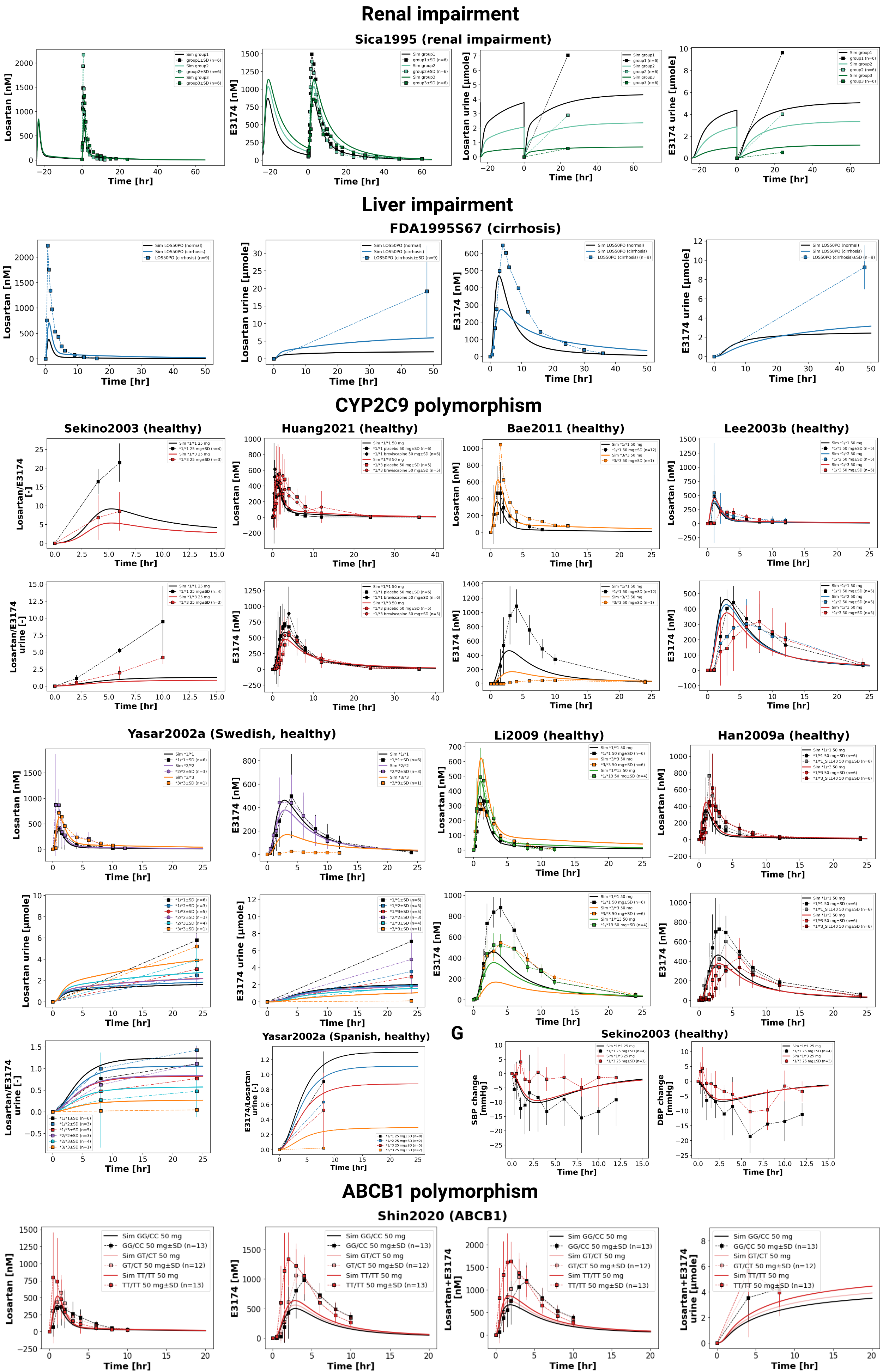
Figure 3:Reproduction of study simulations (renal and liver impairment, CYP2C9 and ABCB1 polymorphism) from the primary publication. Data is taken from Bae et al., 2011FDA, 1995Han et al., 2009Huang et al., 2021Lee et al., 2003Li et al., 2009Sekino et al., 2003Shin et al., 2020Sica et al., 1995Yasar et al., 2002.
4.2Reproduction of Simulations, Experiments, and Scans¶
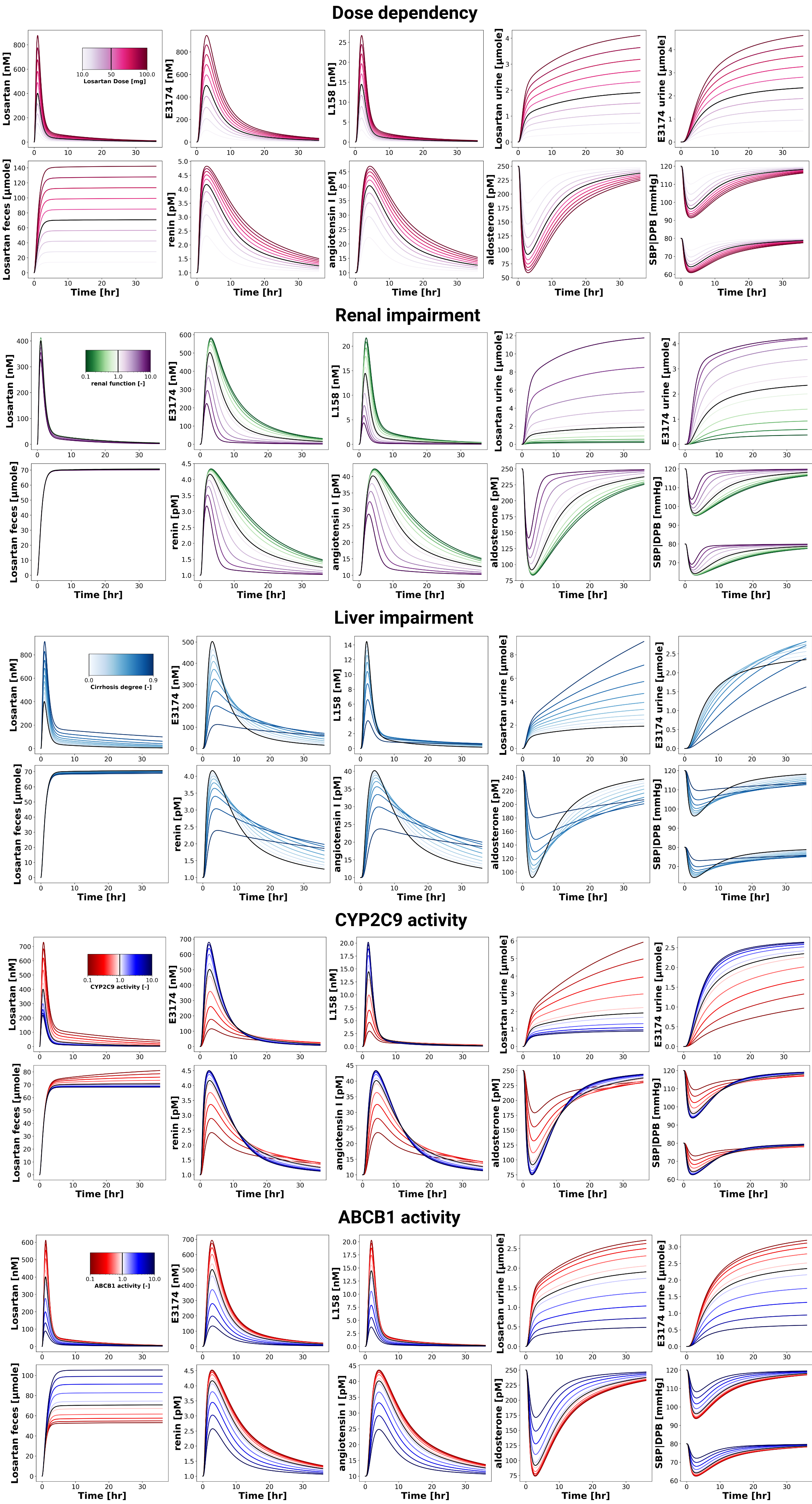
Figure 4:Reproduction of simulation experiments (dose dependency, renal and liver impairment, CYP2C9 and ABCB1 activity) from the primary publication.
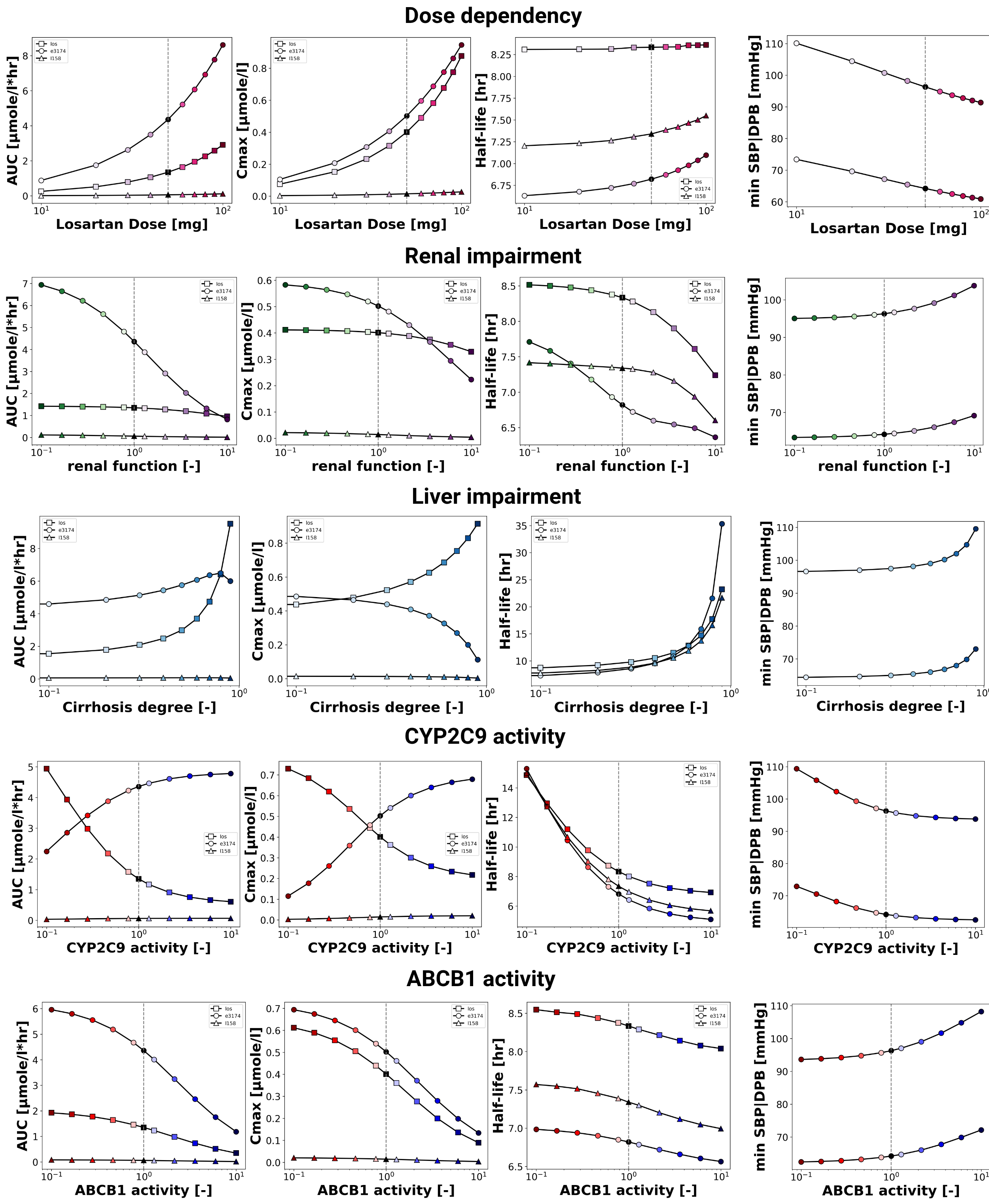
Figure 5:Reproduction of parameter scans (dose dependency, renal and liver impairment, CYP2C9 and ABCB1 activity) from the primary publication.
5Discussion¶
We have demonstrated the computational reproducibility of the key findings from the losartan PBPK/PD model presented in the primary publication. Using the provided simulation scripts, all figures were regenerated without modifying parameters or structure, verifying the consistency of the model. Reproducibility was confirmed across different operating systems using both a local installation with uv and a Dockerized workflow. The uv-based approach allows users to install the package and dependencies natively. In addition, the containerised workflow provides a fully preconfigured environment and ensures consistent results independent of the local setup. Encoding the model in SBML with hierarchical composition removes ambiguity and allows modular reuse of the submodels. Together with the use of community standards and FAIR practices, this provides a transparent and reusable resource that can be applied or extended in future pharmacokinetic/pharmacodynamic modeling work.
Author Contributions¶
E.T. and M.K. contributed to conceptualisation, methodology, data curation, and development of the PBPK/PD model. E.T., M.M., M.E., and M.K. contributed to analyses, software, and visualisation. M.M., M.E., and M.K. contributed to the reproducibility of the computational workflow. M.M. wrote the original draft. E.T., M.M., M.E., and M.K. contributed to the manuscript review and editing. M.K. provided supervision throughout the project. All authors approved the final manuscript.
Funding¶
Matthias König (MK) was supported by the Federal Ministry of Research, Technology and Space (BMFTR, Germany) within ATLAS by grant number 031L0304B and by the German Research Foundation (DFG) within the Research Unit Program FOR 5151 "QuaLiPerF (Quantifying Liver Perfusion-Function Relationship in Complex Resection - A Systems Medicine Approach)" by grant number 436883643 and by grant number 465194077 (Priority Programme SPP 2311, Subproject SimLivA). This work was supported by the BMBF-funded de.NBI Cloud within the German Network for Bioinformatics Infrastructure (de.NBI) (031A537B, 031A533A, 031A538A, 031A533B, 031A535A, 031A537C, 031A534A, 031A532B). Mariia Myshkina was supported by the Federal Ministry of Research, Technology and Space (BMFTR, Germany) within ATLAS by grant number 031L0304B and by the German Research Foundation (DFG) within the Priority Programme SPP 2311, Subproject SimLivA by grant number 465194077. Michelle Elias was supported by the German Research Foundation (DFG) within the Priority Programme SPP 2311, Subproject SimLivA by grant number 465194077.
Acknowledgments¶
Figures were created in BioRender. König, M. (2026) https://
- Lo, M. W., Goldberg, M. R., McCrea, J. B., Lu, H., Furtek, C. I., & Bjornsson, T. D. (1995). Pharmacokinetics of Losartan, an Angiotensin II Receptor Antagonist, and Its Active Metabolite EXP3174 in Humans. Clinical Pharmacology and Therapeutics, 58(6), 641–649. 10.1016/0009-9236(95)90020-9
- Sica, D. A., Gehr, T. W. B., & Ghosh, S. (2005). Clinical Pharmacokinetics of Losartan. Clinical Pharmacokinetics, 44(8), 797–814. 10.2165/00003088-200544080-00003
- Pedro, A. A., Gehr, T. W., Brophy, D. F., & Sica, D. A. (2000). The Pharmacokinetics and Pharmacodynamics of Losartan in Continuous Ambulatory Peritoneal Dialysis. Journal of Clinical Pharmacology, 40(4), 389–395. 10.1177/00912700022009099
- Sica, D. A., Lo, M. W., Shaw, W. C., Keane, W. F., Gehr, T. W., Halstenson, C. E., Lipschutz, K., Furtek, C. I., Ritter, M. A., & Shahinfar, S. (1995). The Pharmacokinetics of Losartan in Renal Insufficiency. Journal of Hypertension. Supplement : Official Journal of the International Society of Hypertension, 13(1), S49-52. 10.1097/00004872-199507001-00007
- Yoshitani, T., Yagi, H., Inotsume, N., & Yasuhara, M. (2002). Effect of Experimental Renal Failure on the Pharmacokinetics of Losartan in Rats. Biological & Pharmaceutical Bulletin, 25(8), 1077–1083. 10.1248/bpb.25.1077
- McIntyre, M., Caffe, S. E., Michalak, R. A., & Reid, J. L. (1997). Losartan, an Orally Active Angiotensin (AT1) Receptor Antagonist: A Review of Its Efficacy and Safety in Essential Hypertension. Pharmacology & Therapeutics, 74(2), 181–194. 10.1016/s0163-7258(97)82002-5
- Fischer, T. L., Pieper, J. A., Graff, D. W., Rodgers, J. E., Fischer, J. D., Parnell, K. J., Goldstein, J. A., Greenwood, R., & Patterson, J. H. (2002). Evaluation of Potential Losartan-Phenytoin Drug Interactions in Healthy Volunteers. Clinical Pharmacology and Therapeutics, 72(3), 238–246. 10.1067/mcp.2002.127945
- Göktaş, M. T., Pepedil, F., Karaca, Ö., Kalkışım, S., Cevik, L., Gumus, E., Guven, G. S., Babaoglu, M. O., Bozkurt, A., & Yasar, U. (2016). Relationship between Genetic Polymorphisms of Drug Efflux Transporter MDR1 (ABCB1) and Response to Losartan in Hypertension Patients. European Review for Medical and Pharmacological Sciences, 20(11), 2460–2467.
- Haufroid, V. (2011). Genetic Polymorphisms of ATP-binding Cassette Transporters ABCB1 and ABCC2 and Their Impact on Drug Disposition. Current Drug Targets, 12(5), 631–646. 10.2174/138945011795378487
- Sekino, K., Kubota, T., Okada, Y., Yamada, Y., Yamamoto, K., Horiuchi, R., Kimura, K., & Iga, T. (2003). Effect of the Single CYP2C9*3 Allele on Pharmacokinetics and Pharmacodynamics of Losartan in Healthy Japanese Subjects. European Journal of Clinical Pharmacology, 59(8–9), 589–592. 10.1007/s00228-003-0664-5
- Shin, H.-B., Jung, E. H., Kang, P., Lim, C. W., Oh, K.-Y., Cho, C.-K., Lee, Y. J., Choi, C.-I., Jang, C.-G., Lee, S.-Y., & Bae, J.-W. (2020). ABCB1 c.2677G>T/c.3435C>T Diplotype Increases the Early-Phase Oral Absorption of Losartan. Archives of Pharmacal Research, 43(11), 1187–1196. 10.1007/s12272-020-01294-3
- Yasar, U., Forslund-Bergengren, C., Tybring, G., Dorado, P., Llerena, A., Sjöqvist, F., Eliasson, E., & Dahl, M.-L. (2002). Pharmacokinetics of Losartan and Its Metabolite E-3174 in Relation to the CYP2C9 Genotype. Clinical Pharmacology and Therapeutics, 71(1), 89–98. 10.1067/mcp.2002.121216
- Grzegorzewski, J., Brandhorst, J., Green, K., Eleftheriadou, D., Duport, Y., Barthorscht, F., Köller, A., Ke, D. Y. J., De Angelis, S., & König, M. (2021). PK-DB: Pharmacokinetics Database for Individualized and Stratified Computational Modeling. Nucleic Acids Research, 49(D1), D1358–D1364. 10.1093/nar/gkaa990
- Hucka, M., Bergmann, F. T., Chaouiya, C., Dräger, A., Hoops, S., Keating, S. M., König, M., Novère, N. L., Myers, C. J., Olivier, B. G., Sahle, S., Schaff, J. C., Sheriff, R., Smith, L. P., Waltemath, D., Wilkinson, D. J., & Zhang, F. (2019). The Systems Biology Markup Language (SBML): Language Specification for Level 3 Version 2 Core Release 2. Journal of Integrative Bioinformatics, 16(2). 10.1515/jib-2019-0021
- Keating, S. M., Waltemath, D., König, M., Zhang, F., Dräger, A., Chaouiya, C., Bergmann, F. T., Finney, A., Gillespie, C. S., Helikar, T., Hoops, S., Malik-Sheriff, R. S., Moodie, S. L., Moraru, I. I., Myers, C. J., Naldi, A., Olivier, B. G., Sahle, S., Schaff, J. C., … SBML Level 3 Community members. (2020). SBML Level 3: An Extensible Format for the Exchange and Reuse of Biological Models. Molecular Systems Biology, 16(8), e9110. 10.15252/msb.20199110





